Diseases
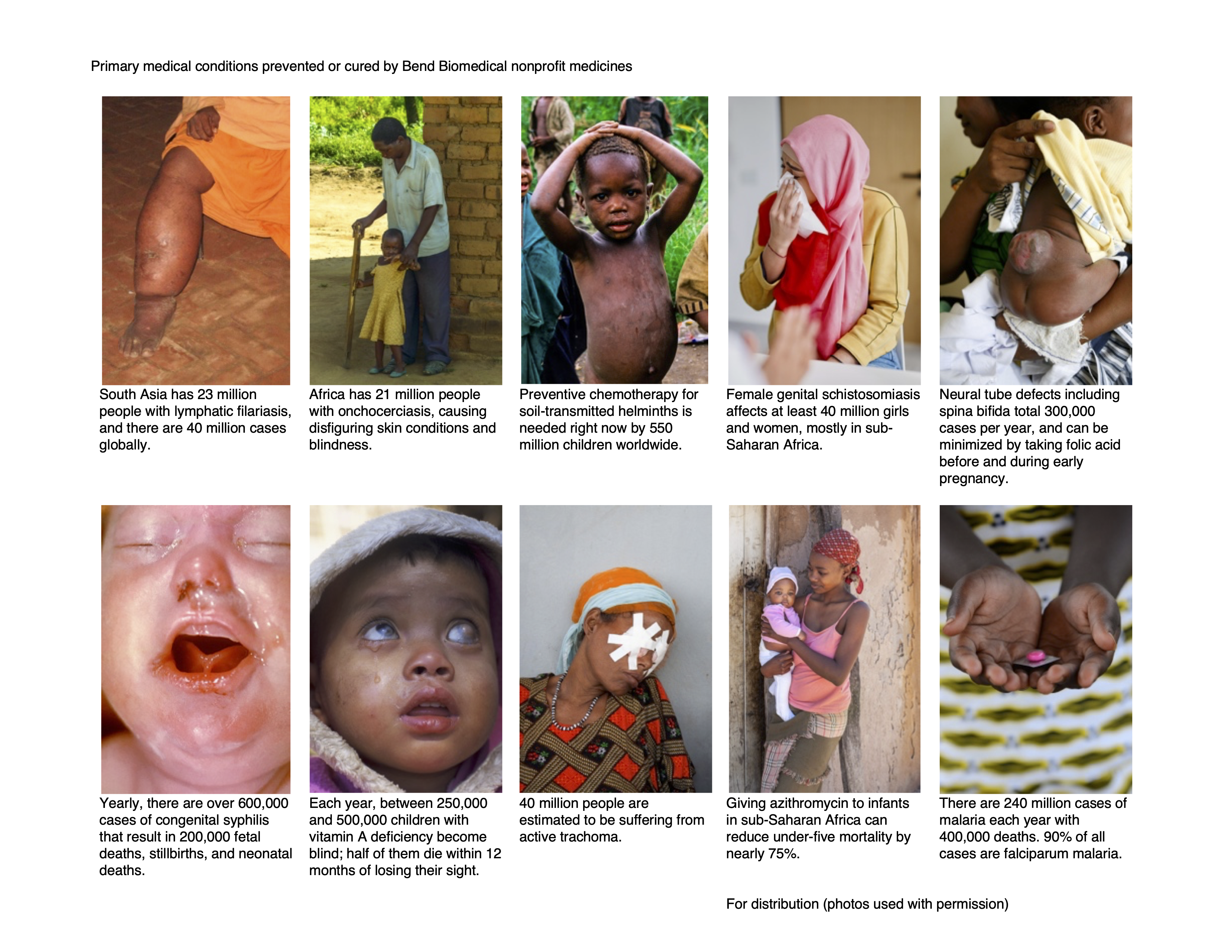
Photo Permissions
Lymphatic filariasis. Photo courtesy Sudesh Kumar.
Onchocerciasis. Photo courtesy Julian Lott.
Soil-transmitted helminths. Photo courtesy Dylan Garcia.
Female genital schistosomiasis. Photo courtesy Cavallini James.
Neural tube defects. Photo courtesy BSIP Scientific and Medical Images.
Congenital syphilis. Photo courtesy Gado Images.
Vitamin A blindness. Photo courtesy Jenny Matthews.
Trachoma. Photo courtesy DMA Alamy.
Under-five mortality prevention. Photo courtesy Lucian Coman.
Falciparum malaria. Photo courtesy Riccardo Lennart Niels Mayer.
Corresponding World Health Organization Guidelines
We participate in a process known as World Health Organization (WHO) prequalification of medicines, a service provided by WHO to assess the quality, safety, and efficacy of medicinal products. Only essential medicines qualify. Countries and organizations now express a preference for WHO prequalified medicines. At Bend Biomedical, we either package in the United States the original name-brand drug, or we use a contract manufacturing organization to manufacture and package a WHO prequalified generic with the same quality and performance as the original drug.
• Guideline: Alternative mass drug administration regimens to eliminate lymphatic filariasis. Geneva: World Health Organization; 2017. 9789241550161-eng.
• Guidelines for stopping mass drug administration and verifying elimination of human onchocerciasis: criteria and procedures. Geneva: World Health Organization; 2016 (updated 2020). 9789241510011-eng.
• Guideline: preventive chemotherapy to control soil-transmitted helminth infections in at-risk population groups. Geneva: World Health Organization; 2017. 9789241550116-eng.
• WHO guideline on control and elimination of human schistosomiasis. Geneva: World Health Organization; 2022. 9789240041608-eng.
• WHO Recommendations on Antenatal Care for a Positive Pregnancy Experience. World Health Organization. January 2017. 9789241549912-eng.
• The Global elimination of congenital syphilis: rationale and strategy for action. Geneva, World Health Organization, 2007. 9789241595858_eng.
• Guideline: Vitamin A supplementation in infants and children 6-59 months of age. Geneva, World Health Organization, 2011. 9789241501767-eng.
• Trachoma Control: A Guide for Programme Managers. Geneva, World Health Organization, 2006. 9789241546904-eng.
• WHO guideline on mass drug administration of azithromycin to children under five years of age to promote child survival. Geneva: World Health Organization; 2020. 9789240009585-eng.
• Mass drug administration for falciparum malaria: a practical field manual. Geneva: World Health Organization; 2017. 9789240009585-eng.

